| FDA 510(k)number | K181691 |
|---|---|
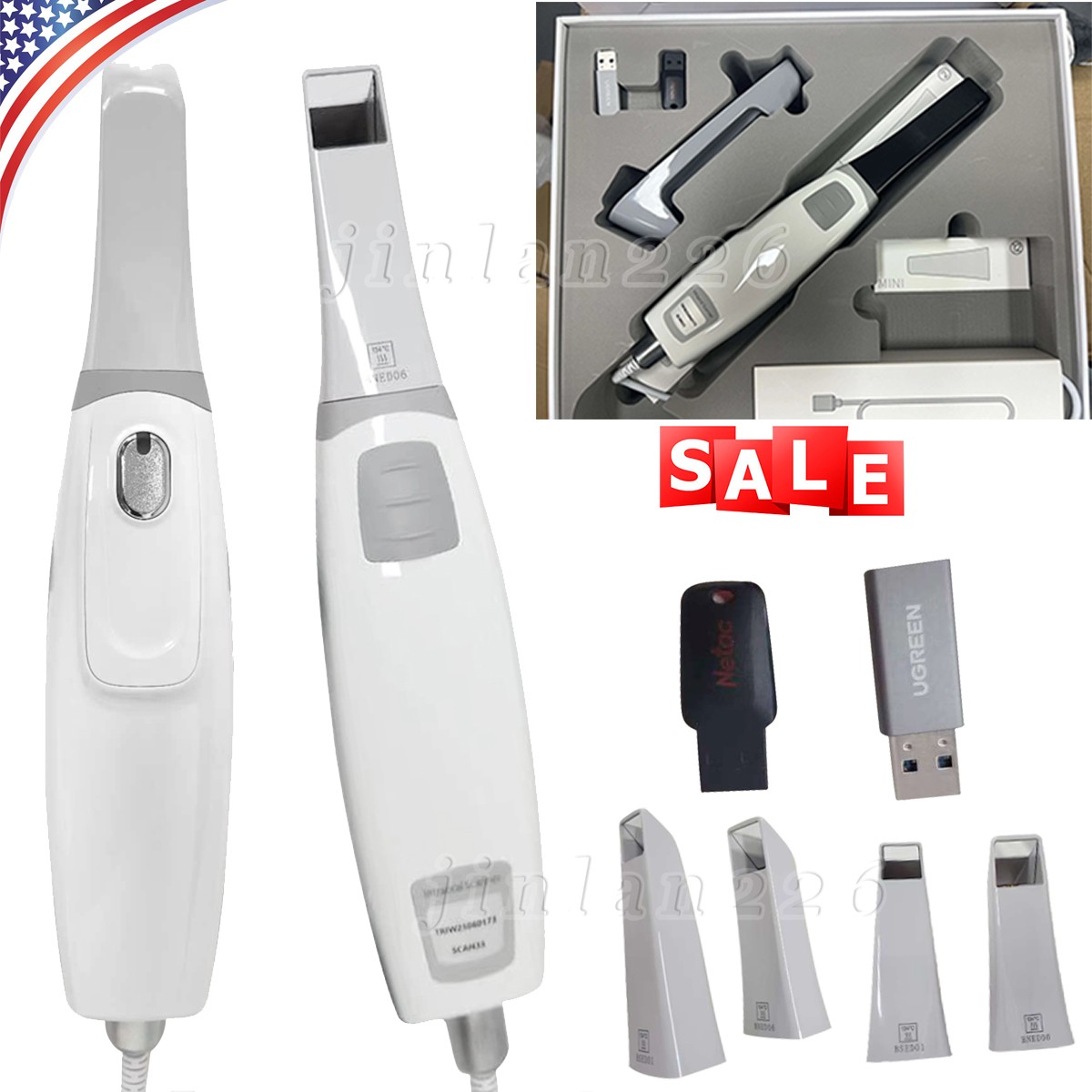
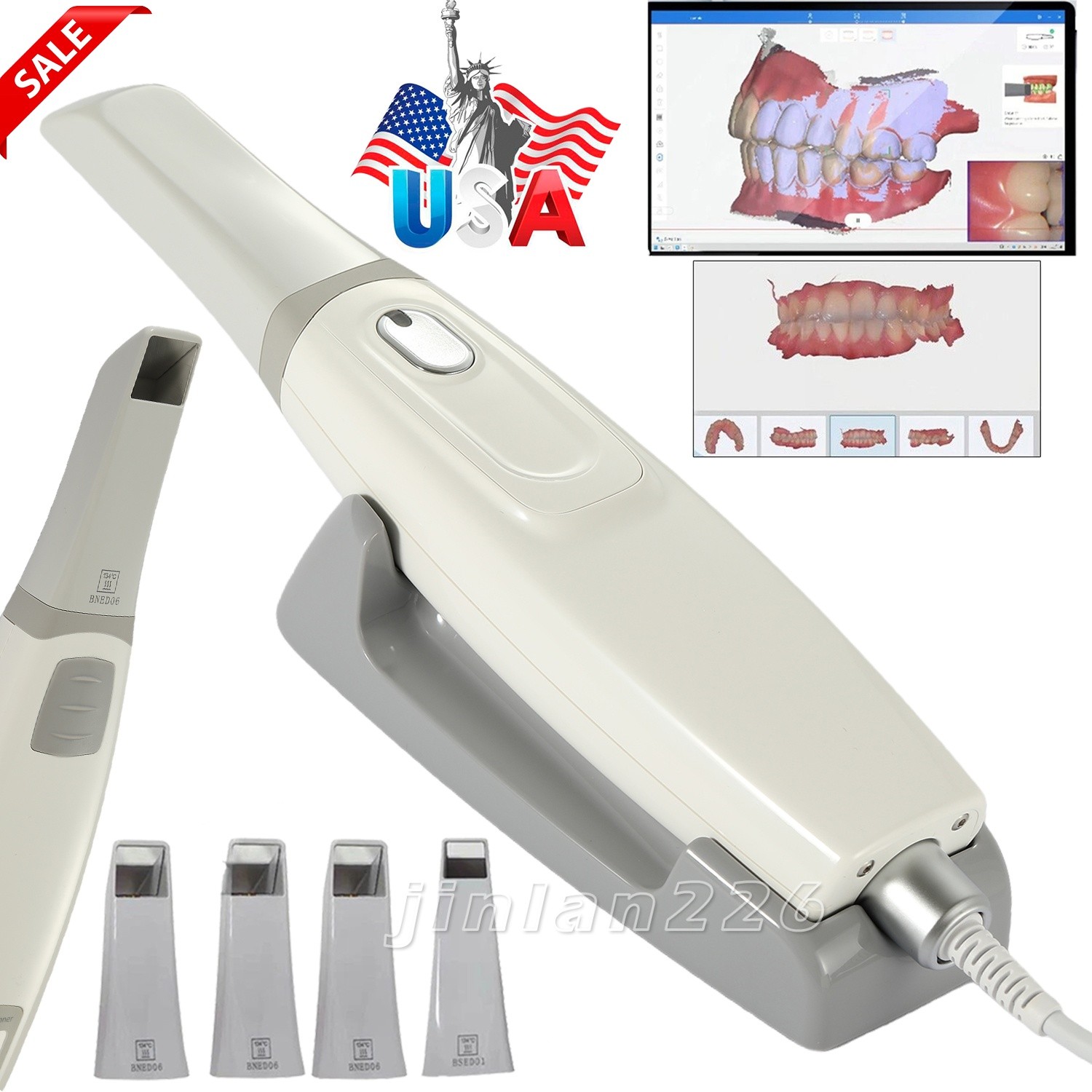
| Scanner Tip | 4 Tips (3 Normal Tip + 1 Smaller Tips) |
| Report | Support Printing Report |
| 510(K) Number For Scaler | K163414 |
| Custom Bundle | No |
| UPC | 612292520274 |
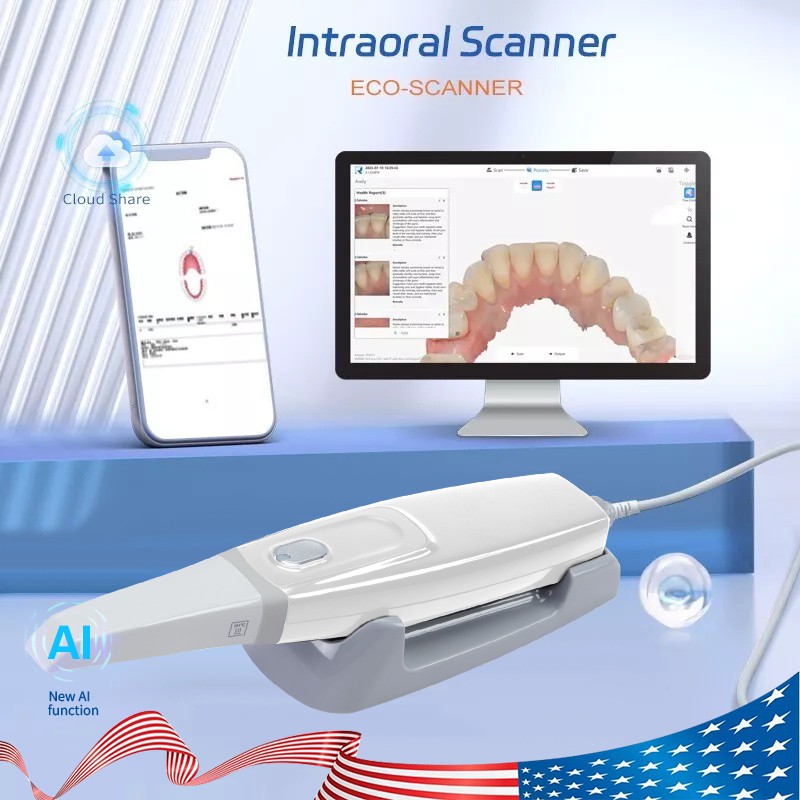
| Product | Acquisition Scanner |
| Export File | STL, PLY, OBJ |
| Languages | Multilingual System (15 Languages) |
| Subsequent Product Code for dental handpiece | EGS |
| Materials sourced from | China |
| Modified Item | No |
| Intended Use/Discipline | Dental Laboratory, Dentistry |
| Brand | SANDENT |
| 510(K) Number For Dental Handpiece | K181691 |
| Accuracy | Single crown≤10um, single arch≤15um, full arch≤20um |
| Classification Product Code for dental handpiece | EFB |
| 510(K) Number For Curing Light | K192233 |
| Model | Scan 33 |
| Regulation Number for dental handpiece | 872.4200 |
| Software | Free Software (Free Updated) |
Check the listing for details. Dental Digital Imaging Scanner for Restorations, Implants 3D Intraoral Scanner. Condition: New. Listed at 2958.00 USD. FDA for Curing Light: 510(K) Number: K192233, Regulation Number: 21 CFR 872.6070, Product Code: EBZ. FDA for Handpiece: 510(K) Number: K181691, Regulation Number: 872.4200, Classification Product Code: EFB, Subsequent Product Code: EGS.