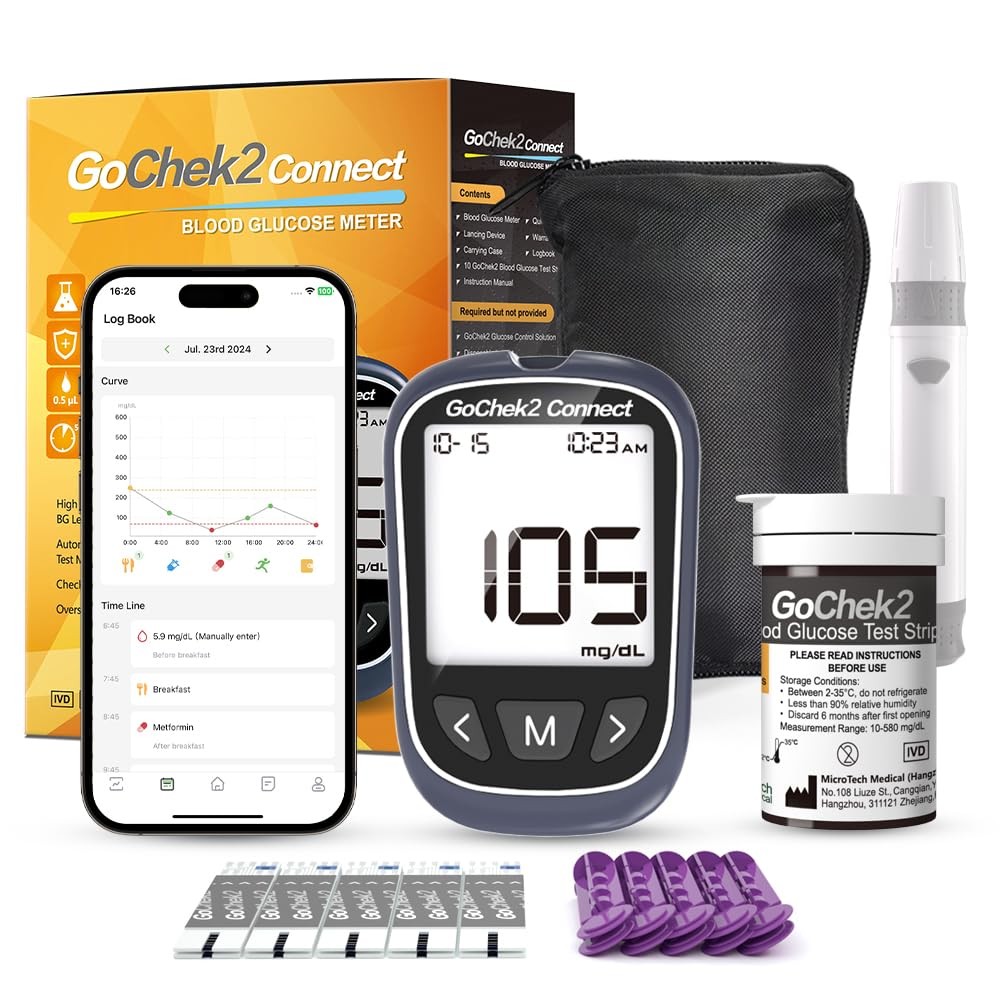
| Included Components | Blood Monitor Kit, Lancet, Test Strip |
|---|---|
| Fda Premarket Approval Number Pma | 510K# (Market Pre-Notification#) K109943 |
| Model Name | Gocheck2 Bluetooth |
| Fda Device Classification | Ii |
| Fda Indication Of Use | Otc |
| Operating Time | 5.0 |
| Part Number | Microtech |
| Fda Instructions For Use Type | Consumer |
| Battery Weight | 2.5 |
| Number Of Lithium Metal Cells | 2 |
| Model Number | Bluetooth 10Kit |
| Fda Label Type | Consumer |
| Batteries Required? | Yes |
| Batteries Included? | Yes |
| Fda Premarket Approval Number 510K | 510K# (Market Pre-Notification#) K109943 |
| EAN | 6958590309154 |
| ISBN | 6958590309154 |
| Package Dimensions LxWxH | 7.13x6.73x1.85 Inches |
| Weight | 0.51 Pounds |
Check the listing for details. MicroTech Bluetooth Blood Sugar Testing Kit 10 Strips Lancets, Free App, No .... Condition: New. Listed at 13.94 USD. It has obtained certifications such as FDA, CE, GMP, and ISO 15197:2015. 【Get a quick and gentle test in 5 seconds】: The Microtech blood sugar test kit is designed for comfort. The high precision blood sugar detection system only requires a tiny 0.5µl blood sample and can give accurate results in 5 seconds.