| Country of Origin | China |
|---|---|
| FDA for Curing Light | 510(K) Number: K192233 |
| FDA for Handpiece | 510(K) Number: K181691 |
| FDA for Scaler | 510(K) Number: K163414 |
| Custom Bundle | No |
| Product Code | EBZ |
| 510(K) Number | K192233 |
| Regulation Number | 21 CFR 872.6070 |
| Modified Item | No |
| Intended Use/Discipline | Dental Laboratory, Dentistry |
| Brand | SANDENT |
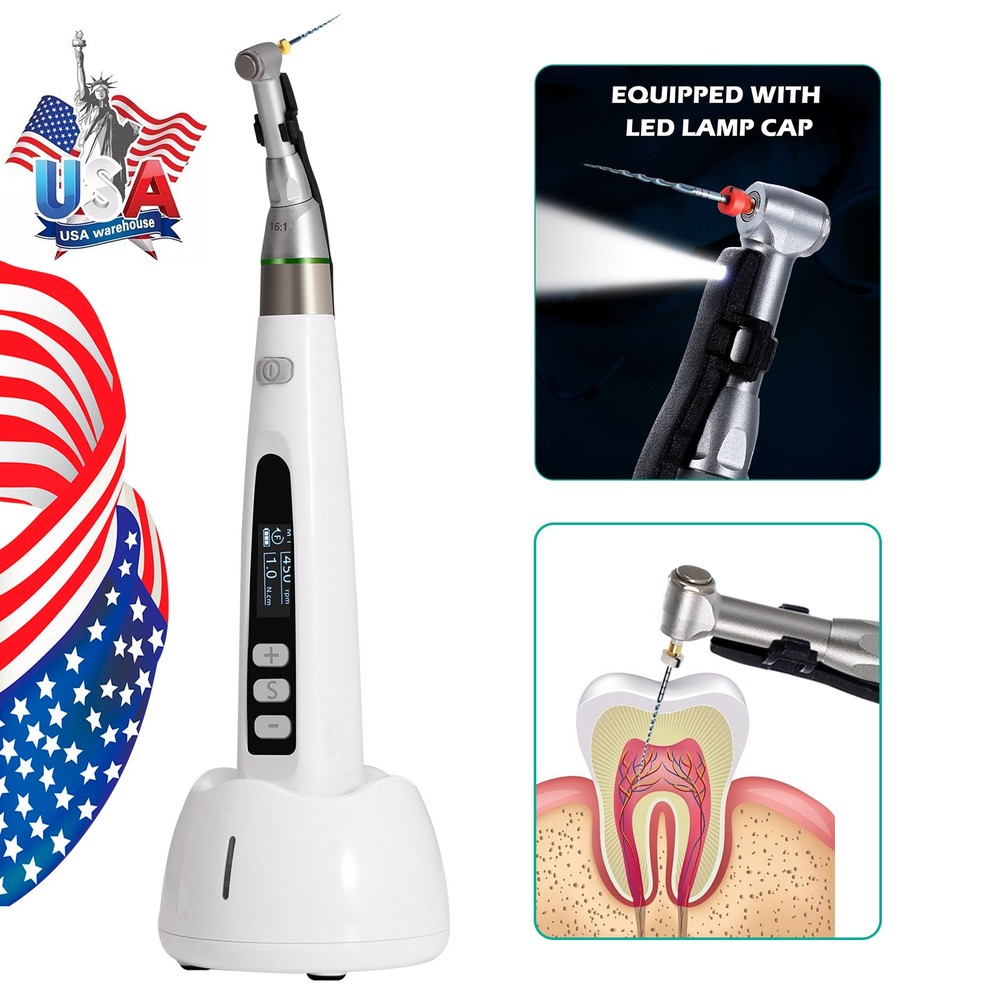
Check the listing for details. Wireless Endo Motor Handpiece LCD Display Auto Reverse Portable. Condition: New, Made in China. Listed at 66.99 USD. (It is mainly for your piece of mind and comfort.). Subsequent Product Code: EGS. Classification Product Code: EFB. Regulation Number: 872.4200. 510(K) Number: K181691.











